Antimicrobial Plus Hospital Curtains - Standard Colours
Why Antimicrobial Plus?
Unlike other available antimicrobial curtains, Endurocide® Antimicrobial Plus Curtains are:
| Proven to remain active for up to two years* | ||
| Silver additive free |
||
| Tested and proven to be effective against the top five pathogen groups found in hospitals: bacteria, fungi, mycobacteria, spores & viruses* | ||
Other important benefits:
 |
 |
 |
 |
|||
| PATENTED COATING for up to two year long life* |
REDUCING RISK & saving valuable time |
OVERALL SAVINGS over two years |
PEER REVIEW TESTED in over eight countries |
International Peer Reviews
| Hospital COVID-19 report | ||
| NHS Hospital Trust report | Published article |
*Disclaimer: Two year long life applies to Endurocide® Antimicrobial Plus Standard Curtains only and whilst Endurocide® Antimicrobial Plus Standard Curtains have been independently tested to remain antimicrobially and sporicidally effective for up to two years in-situ, the actual length of curtain use achieved will depend on a variety of factors, including, but not limited to: individual hospital practices; the natural longevity of polypropylene; the risk of the curtain being soiled from items such as blood, urine and general spills; etc. Curtains should always be replaced when visibly soiled. Any timescales provided/referenced are always offered as a guideline only and under no circumstances whatsoever constitute a guarantee. See individual curtain testing pages for full details on pathogens tested against.
International Laboratory Testing
- Sporistatic, bacteriostatic, fungostatic and mycobacteriostatic testing
- Sporicidal, bactericidal & fungicidal testing
- Liquid treatment testing
- Fire retardancy testing
To validate our claims, Endurocide® Antimicrobial Plus Curtains have been independently laboratory tested to international standards against pathogens including:
| C.difficile spores | Serratia marcescens | MRSA | |||||
| VRE | E.coli | E.hirae | |||||
| Pseudomonas aeruginosa |
ESBL E.coli |
ESBL Klebsiella pneumoniae | |||||
| Salmonella choleraesuis |
Acinetobacter baumanii |
Mycobacteria tuberculosis | |||||
| Aspergillus niger |
Candida albicans |
Vaccinia virus |
|||||
| Human Coronavirus |
Measles virus |
H1N1 Swine flu |
Sporistatic & Bacteriostatic testing
Qualitative “Zone of Inhibition” tests demonstrate that the fabric is sporistatic and bacteriostatic and whether it can prevent pathogens from growing and reproducing on treated fabric. The International Standards for bacteriostatic & sporistatic fabrics are ISO 20645, AATCC 147 and CG 147.
New curtains "box fresh" |
|||
Spores |
Standard |
Standard |
|
|
CG 147 |
|
AATCC 147 |
Bacteria |
Standard |
Standard |
|
|
CG 147 |
|
CG 147 |
|
CG 147 |
|
CG 147 |
|
CG 147 |
|
ISO 20645 |
|
ISO 20645 |
|
CG 147 |
|
CG 147 |
|
ISO 20645 |
|
AATCC 147 |
|
CG 147 |
Fungi |
Standard |
Standard |
|
|
AATCC 147 |
|
CG 147 |
|
CG 147 |
|
CG 147 |
Mycobacteria |
Standard |
||
|
CG 147 | ||
Curtains 1 year after manufacture |
|||
Spores |
Standard |
||
|
AATCC 147 |
|
CG 147 |
Bacteria |
Standard |
Standard |
|
|
AATCC 147 |
|
CG 147 |
|
AATCC 147 |
|
CG 147 |
|
CG 147 |
|
AATCC 147 |
Fungi |
Standard
|
||
|
CG 147 | ||
Curtains 2 years after manufacture |
|||
Bacteria |
Standard |
Standard |
|
|
CG 147 |
|
CG 147 |
|
CG 147 |
|
CG 147 |
|
CG 147 |
|
CG 147 |
Sporicidal & Bactericidal testing
Quantitative tests to demonstrate that the treated fabric is capable of killing pathogens. The International Standards for biocidal treated fabrics are ISO 20743, ATCC 100 and JIS Z 2801.
New curtains "box fresh" |
|||
Spores |
Standard |
Standard |
|
|
AATCC 100 |
|
JIS Z 2801 |
Bacteria |
Standard |
Standard |
|
|
AATCC 100 |
|
AATCC 100 |
|
JIS Z 2801 |
|
JIS Z 2801 |
|
AATCC 100 |
|
AATCC 100 |
|
ISO 20743 |
|
ISO 20743 |
|
JIS Z 2801 | ||
Fungi |
Standard |
Standard |
|
|
AATCC 100 |
|
AATCC 100 |
|
AATCC 100 |
|
JIS Z 2801 |
Curtains 1 year after manufacture |
|||
Spores |
Standard |
||
|
AATCC 100 | ||
Curtains 2 years after manufacture |
|||
Spores |
Standard |
Standard |
|
|
AATCC 100 | ||
Liquid treatment testing
The following testing has also been carried out on the liquid used to provide the antimicrobial and sporicidal coating on the curtains:
Enveloped viruses |
Standard |
Standard |
|
|
EN 14476 |
|
EN 14476 |
|
EN 14476 |
|
EN 14476 |
Fire Retardant Standards
Our curtains have been tested to the following International fire retardant standards:
Country/Region |
Standard |
||||||
|
EN 13773: 2003+A1: 2007 BS 5867 Part 2 Types B & C: 2008 |
||||||
|
CAN/ULC-S109; NFPA 701: 2010 | ||||||
|
AS 2755.2: 1985 AS 1530.2-1993 Part 2 |
||||||
For further information or to request copies of our test certificates, please contact us.
* DISCLAIMER: Whilst Endurocide® Plus Curtains have been independently tested to remain antimicrobially and sporicidally effective for up to two years in-situ, the actual length of curtain use achieved will depend on a variety of factors, including, but not limited to: individual hospital practices; the natural longevity of polypropylene; the risk of the curtain being soiled from items such as blood, urine and general spills; etc. Curtains should always be replaced when visibly soiled. Any timescales provided/referenced are always offered as a guideline only and under no circumstances whatsoever constitute a guarantee.
Product Options
Endurocide® Antimicrobial Plus Curtains are available in a number of different types and colours and with different hooks to suit your requirements.
Curtain types
StandardFor use with suspended ceiling rails. |
Mesh TopWith NFPA 13 compliant mesh for sprinkler access with ceiling-fixed rails. |
Mesh Cut†An economical alternative to the Mesh Top curtain. |
Long DropFor use with ceiling-fixed rails. |
|||
 |
 |
 |
 |
Colours

Hanging Systems
Eyelet only |
Quick-fit hooks
|
Large-top hooks
|
U-type hooks |
|||
 |
 |
 |
 |
|||
Wheeled hooks |
Wide-wheeled hooks
|
Metal-bar wide-wheeled hooks
|
Eyelet extension hooks |
|||
 |
 |
 |
 |
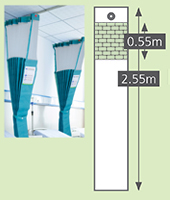
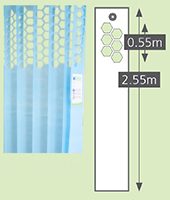
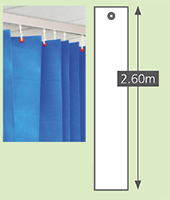
Sizes
|
|
Standard |
Long drop |
Meshtop |
Meshcut |
|
2.0m | 2.6m |
2.55m |
2.55m |
|
7.5m |
7.5m |
7.5m |
7.5m |
|
5.55m |
5.55m |
5.55m |
5.55m |
|
3.75m |
3.75m |
3.75m |
3.75m |
|
N/A | N/A | 0.55m | 0.55m |
As a general rule, total curtain width needs to be 2 x length of curtain rail. . Please note there is a +/-5% tolerance on all aspects of the curtains. |
||||
Order Code Selector
To generate an Order Code, please use our handy online Code Selector tool:
† Note: Meshcut curtains do not meet US NFPA 13 specifications and are not suitable in areas that require mesh for sprinkler access.
Our technology is different...
Whilst traditional polyester curtains, natural fibre or short-life disposable curtains can easily become sources of pathogenic transmission within days of installation, our curtains are different...
|
|
At the curtain manufacturing stage, the polypropylene curtain fabric is impregnated with our unique, patented Endurocide® Curtain Liquid. This impregnation coats the curtain fabric creating a polymer layer which has a dual ‘static’ and ‘cidal’ action. The ‘static’ action allows the curtain fabric to trap pathogens on the surface of the fabric and prevent them from multiplying, whilst the ‘cidal’ action then kills the pathogens - helping to break the chain of infection! |
|
Breaking the chain of infection
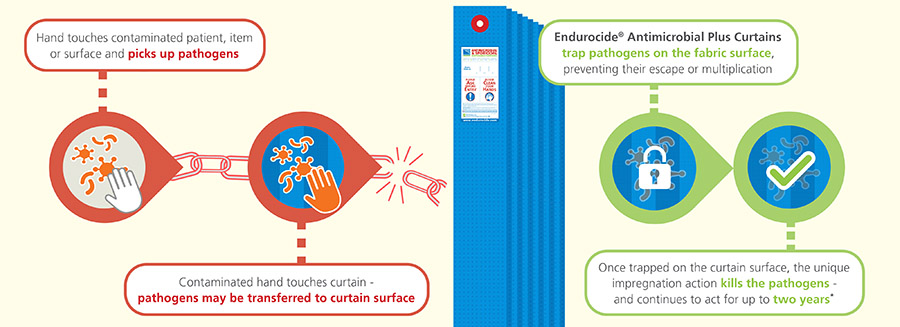
Endurocide® Antimicrobial Plus Curtains prevent potential retransmission of pathogens to staff, patients, visitors, equipment, surfaces and any other items - breaking the chain of infection!
Our technology in action...
Endurocide® Antimicrobial Plus Curtains kill the top 5 pathogen groups:*
 |
 |
 |
*Disclaimer: Two year long life applies to Endurocide® Antimicrobial Plus Standard Curtains only and whilst Endurocide® Antimicrobial Plus Standard Curtains have been independently tested to remain antimicrobially and sporicidally effective for up to two years in-situ, the actual length of curtain use achieved will depend on a variety of factors, including, but not limited to: individual hospital practices; the natural longevity of polypropylene; the risk of the curtain being soiled from items such as blood, urine and general spills; etc. Curtains should always be replaced when visibly soiled. Any timescales provided/referenced are always offered as a guideline only and under no circumstances whatsoever constitute a guarantee. See individual curtain testing pages for full details on pathogens tested against.
In the UK:
Hospitals: |
Bulk purchase or reseller information: |
|
Outside the UK:
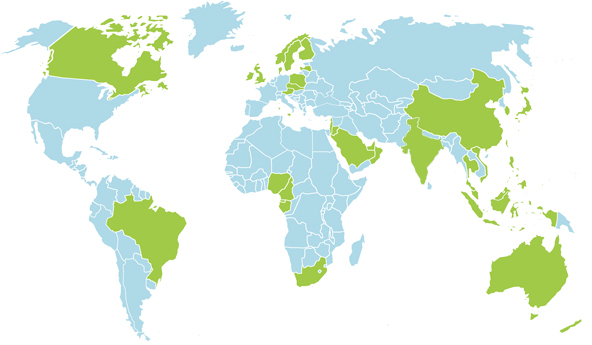
We have existing Distributors in the countries highlighted in green - for information on who to contact in your territory, please contact us.
Interested in becoming a Distributor?
If you'd like to find out how to become our distributor in any other territory, we would be very pleased to hear from you!
How do Endurocide® Antimicrobial Plus Hospital Curtains work?
At the curtain manufacturing stage, the polypropylene curtain fabric is impregnated with our unique, patented Endurocide® Curtain Liquid (which is a water-based formulation, based on a combination of active quaternary ammonium compounds) and a flame retardant chemical.
The Endurocide® Curtain Liquid coats the fabric with a biostatic and biocidal polymer layer. If pathogens come into contact with the fabric, the Endurocide® Curtain Liquid layer instantly starts working by trapping the pathogens on the fabric and preventing them from multiplying and being re-transmitted.
 |
|
Why were Endurocide® Antimicrobial Plus Hospital Curtains created?
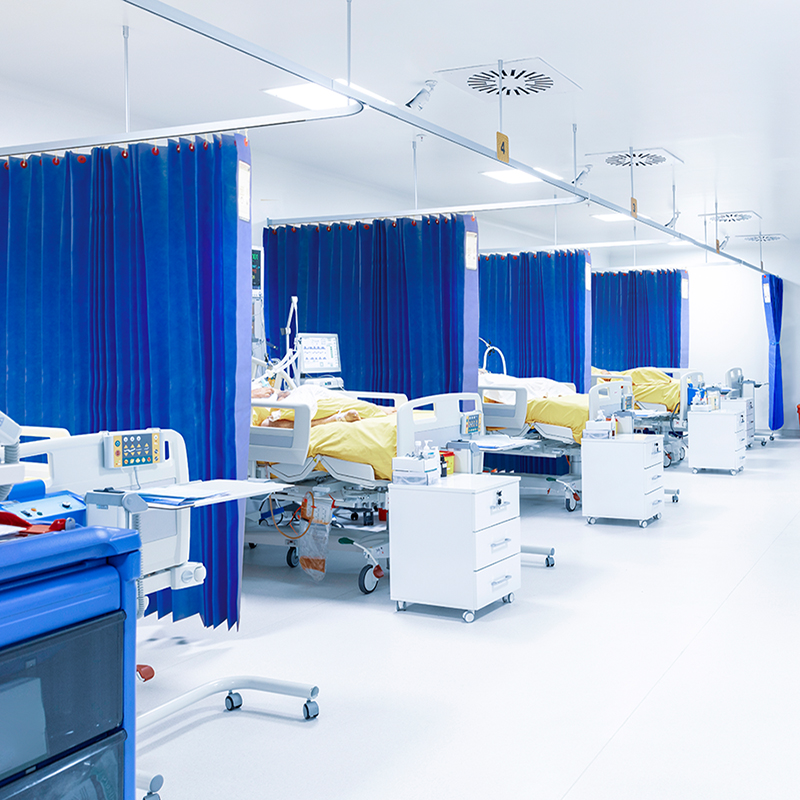
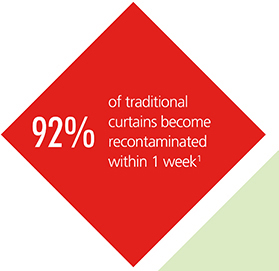
Studies have shown that traditional hospital “curtains are rapidly contaminated with potentially pathogenic bacteria” and “curtains are a potentially important site of bacterial contamination in hospitals”1.
On top of this, further studies show that pathogens such as MRSA, VRE and C.difficile spores are capable of surviving on surfaces for weeks, meaning that hospital workers can touch these contaminated surfaces and then transmit these pathogens onto patients2.
Traditional polyester curtains, or standard polypropylene curtains that haven’t been treated with an antimicrobial agent, can quickly & easily become sources of pathogenic transmission. For example, a healthcare worker who touches the patient could possibly transmit this infection to an object/surface, such as a hospital curtain. If this happens, this object now has the potential to contaminate anyone who comes in contact with the surface. All of this means that traditional Hospital Curtains are a potential source for the spread of healthcare-associated pathogens3.
Endurocide® Antimicrobial Plus Hospital Curtains were specifically created to help hospitals reduce the potential for this pathogenic transmission to occur from Hospital Curtains and they are therefore effective against the top pathogen groups commonly found in hospitals worldwide:
|
|
|
|
|
|
||
Sources:
-
Ohl, Michael; Schweizer, Marin; Graham, Maggie; Heilmann, Kristopher; Boyken, Linda; Diejema, Daniel “Hospital privacy curtains are frequently and rapidly contaminated with potentially pathogenic bacteria” American Journal of Infection Control (2012)
-
Boyce, John M “Environmental contamination makes an important contribution to hospital infection” Journal of Hospital Infection (2007) 65(S2) 50-54
-
Trillis III, Floyd; Eckstein, Elizabeth C; Budavich, Rachel; Pultz, Michael J; Donskey, Curtis J “Contamination of Hospital Curtains with Healthcare-Associated Pathogens” Infection Control and Hospital Epidemiology (November 2008) Vol. 29, No.11
What makes Endurocide® Antimicrobial Plus Hospital Curtains different to other curtains?
Traditional curtains have the potential to become sources of transmission for Hospital Acquired Infections (HAIs). To counteract this, in 2005 we created a disposable curtain that offered antimicrobial protection.
However, the major flaw with the antimicrobial system is that it is unable to have an effect against spores such as C.difficile, which can survive on surfaces for very long periods of time. This means that even antimicrobial curtains can have the potential to become a source of transmission of HAIs within the hospital.
Endurocide® Antimicrobial Plus Hospital Curtains are different to existing curtains on the market because they have been specifically designed to help break the chain of infection and help prevent the spread of HAIs in the hospital, offering a safe to use, long-lasting and powerful curtain capable of killing bacteria, fungi, spores, mycobacteria and enveloped viruses.
Using a patented formula, which is only available on Endurocide® Antimicrobial Plus Hospital Curtains, the curtains become an effective barrier to the transmission of infections, by trapping and killing pathogens on the curtain surface in seconds!
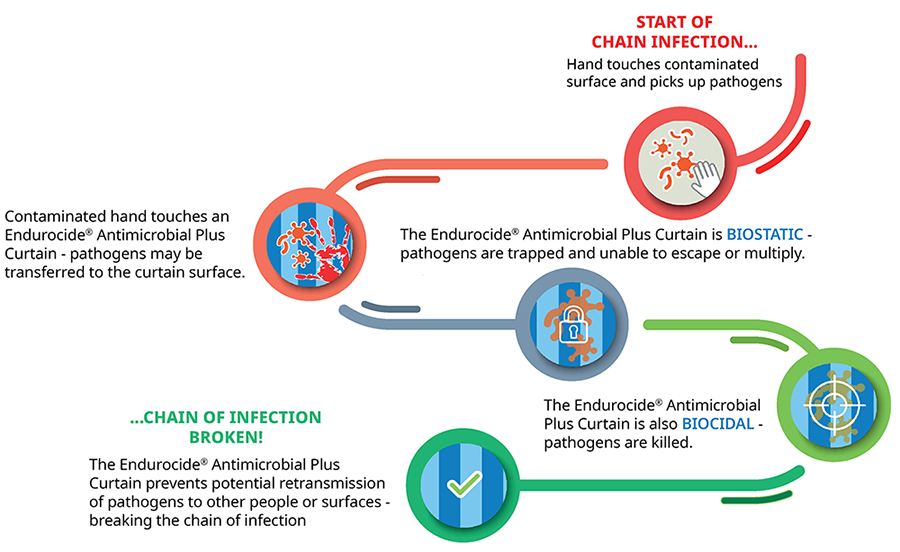
This dual mechanism of trapping & killing the pathogens is key. The Curtain fabric traps pathogens on the surface, preventing them from being picked up when the Curtain is touched and then, when the pathogens are trapped, the Curtain kills the pathogens, preventing them from multiplying or remaining a risk to users on the Curtain.
How can Endurocide® Antimicrobial Plus Hospital Curtains help reduce the spread of HAIs?
Studies have shown that up to 40% of HAIs can be attributed to the cross infection from the hands of health care personnel, who are contaminated either by direct contact with a patient or indirectly by touching contaminated surfaces1. It has also been shown that contaminated hands can transfer viruses to 5 more surfaces2 or 14 other subjects3.
On top of this, pathogens can survive on surfaces for long periods of time, with C.difficile spores and MRSA being shown to survive on surfaces for up to 5 months and 7 months respectively4. It is widely understood that hand hygiene protocols and surface disinfection procedures are essential in minimising the potential for this cross contamination.
However, what about the hospital curtain? What procedures are there to minimise its potential for contamination? If a health care worker touches a contaminated surface and picks up the contamination, then touches a hospital curtain, they have the potential to contaminate the hospital curtain. With some hospital curtains remaining in-situ for anywhere between one day to six months, these hospital curtains have the potential to become sources of transmission of infections for anyone who comes into contact with them.
Until now there was not much that you could do about this. However, Endurocide® Antimicrobial Plus Hospital Curtains have changed this! By being tested and proven to kill spores, bacteria and fungi, Endurocide® Antimicrobial Plus Hospital Curtains offer an effective barrier to pathogens and help hospitals maintain their hand and surface hygiene protocols – by minimising the potential for the hospital curtain to become a source of infection.
-
Weber, David J; Rutala, William A; Miller, Melissa; Huslage, Kirk; Bennet-Sickbert, Emily “Role of hospital surfaces in the transmission of emerging health care-associated pathogens: Norovirus, Clostridium difficile, and Acinetobacter species” Am J Infect Control (2010) 38 S25-33
-
Barker, J; Vipond, IB; Bloomfield, SF “Effects of cleaning and disinfection in reducing the spread of norovirus contamination via environmental surfaces” Journal of Hospital Infection (2004) 58:42-44
-
Von Rheinbaben, F; Schunemann, S; Gross, T; Wolff, MH “Transmission of viruses via contact in a household setting: experiments using bacteriophage strain phiX!!74 as a model virus” Journal of Hospital Infection (2000) 46:61-66
-
Kramer, Axel; Schwebke, Ingeborg and Kampf, Günter “How long do nosocomial pathogens persist on inanimate surfaces? A systematic review” BMC Infectious Diseases (2006), 6:130
Virus testing
Reliable fabric testing against certain viruses, such as Measles and Coronavirus does not yet exist for disposable Hospital Curtains.
Therefore, to demonstrate the virucidal effectiveness of our formulation, we instead use quantitative suspension laboratory tests that are conducted on the liquid used to impregnate our Endurocide® Antimicrobial Plus Curtains.
We are continually adding additional pathogens to our efficacy list, however, we currently have the following Enveloped Virus testing conducted:
|
Pathogen |
Type of Virus |
International Standard |
Comments |
|
Enveloped virus |
EN 14476 |
Vaccinia virus is a large, complex, enveloped virus belonging to the poxvirus family. Successful testing against vaccinia virus is generally considered to allow efficacy claims to be made for all enveloped viruses as it is classed as a reference enveloped virus. This includes claims as to effectiveness against Coronavirus. Vaccinia is also often selected for testing as a substitute for Monkey Pox and other poxviruses, which are difficult to test in laboratories due to their biohazard rating. |
|
Enveloped virus |
EN 14476 |
|
|
Enveloped virus |
EN 14476 |
|
|
Enveloped virus | EN 14476 |
Should you have any questions regarding any of our testing, or wish to discuss anything further, please don’t hesitate to contact us.
How quickly do Endurocide® Antimicrobial Plus Hospital Curtains start working?
Endurocide® Antimicrobial Plus Curtains are ready to work as soon as you take them out of the box and have been independently tested and shown to kill pathogens in seconds.
If you would like more information about the testing that has been conducted on our Endurocide® Antimicrobial Plus Curtains, please visit our Our Technology or International Testing tabs, or contact us for more details on:
|
|
What's the difference between our Curtains and competitor Curtains?
We have found that there are numerous products on the market that claim to be antimicrobial, but do not define what this means and what exactly the product is capable of doing.
When comparing products, it is very important to understand how an antimicrobial hospital curtain works and how its effectiveness is tested and verified. It is also important to understand that there are different ways that an antimicrobial curtain can work, for example:
- Does the curtain simply trap the bacteria on the fabric?
- Does it actually kill the bacteria?
- Does it do both?
Endurocide® Antimicrobial Plus Hospital Curtains are independently tested and shown to not only trap the pathogens on the fabric, but to also kill them - meaning that the curtains are sporistatic, bacteriostatic, sporicidal and bactericidal.
Sporistatic & Bacteriostatic
These terms refer to whether a curtain is capable of inhibiting the growth and reproduction of spores and bacteria. As our Endurocide® Antimicrobial Plus Hospital Curtains have been independently tested and shown to be both Sporistatic and Baceteriostatic, our curtains will trap any spores and bacteria that come into contact with the curtain and prevent these pathogens from escaping and being re-transmitted to another surface.
- The International Standards to demonstrate that a fabric is Sporistatic and Bacteriostatic are AATCC 147 & CG 147. These tests are qualitative tests, in that they evaluate the fabric’s antimicrobiality.
Sporicidal & Bactericidal
This means that the curtain is capable of actually killing the spores and the bacteria on the surface of the curtain, preventing them from multiplying and becoming a source of transmission.
- The International Standards to demonstrate that a fabric is Sporicidal and Bactericidal are ISO 20743, AATCC 100 and JIS Z 2801. These tests are quantitative tests, in that the number/reduction of bacteria on the curtain is counted.
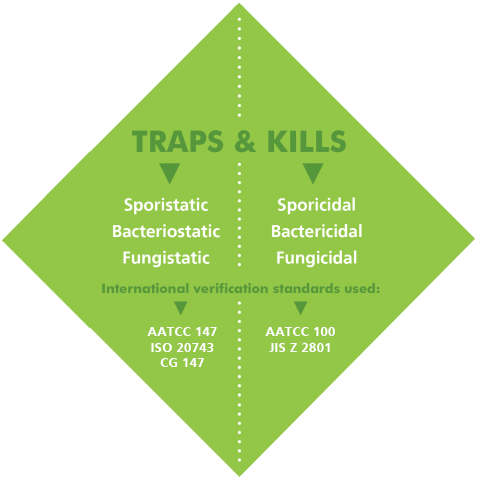
Although we refer to the Endurocide® Antimicrobial Plus Hospital Curtains as being primarily Sporicidal & Antimicrobial, it is important to understand that in order for the curtain to be truly effective, it must have been tested and shown to be both Sporistatic & Bacteriostatic as well as being Sporicidal & Bactericidal.
As far as we are aware, our patented Endurocide® Antimicrobial Plus Hospital Curtains are the only curtains available on the market that can claim this!
Where should you use Endurocide® Antimicrobial Plus Hospital Curtains?
Endurocide® Antimicrobial Plus Curtains have been designed to be used wherever Infection Control & Infection Prevention is of paramount concern.
We would recommend that Endurocide® Antimicrobial Plus Curtains should be used in, amongst others, Intensive Care Wards, High Dependency Units, Isolation Wards, Burn Units, Cardiology Wards, General Surgery Wards, Maternity Wards, Neonatal Wards and Orthopaedic Wards.
Please refer to the table below for more information:
|
Intensive care units |
|
|
Gynaecology |
|
|
High dependency units |
|
|
Obstetrics |
|
|
Infection disease units |
|
|
Isolation wards |
|
|
Burns units |
|
|
Accident and Emergency |
|
|
Cardiology wards |
|
|
General wards |
|
|
Oncology wards |
|
|
Day wards |
|
|
Operation suites |
|
|
General practice wards |
|
|
General surgery wards |
|
|
Occupational therapy |
|
|
Maternity & neonatal wards |
|
|
Physiotherapy |
|
|
Renal units |
|
|
Rheumatology |
|
How do you store Endurocide® Antimicrobial Plus Hospital Curtains?
All Endurocide® Curtains should be stored at ambient temperatures - i.e. at a maximum of 25°C max - and out of direct UV light.
If stored as above, Polypropylene remains stable for approximately 12 months, but after this time will start to gradually degrade. This degradation process will occur at a faster rate in the presence of warm/high temperatures, humid conditions and most importantly in the presence of UV light.
Any timescales provided/referenced are always offered as a guideline only and under no circumstances whatsoever constitute a guarantee.
Are there any instructions supplied with Endurocide® Curtains?
Yes: every box of Endurocide® Curtains includes an Instruction Sheet in order to give our end users clear information on how to use and get the most out of their curtains.
- To download a copy, please click here.
Can I clean or wipe Endurocide® Disposable Curtains?
No: disposable curtains are designed to be replaced when visibly soiled and it is not advised to clean any of our curtains by any mechanical means.
Regarding Endurocide® Antimicrobial Plus Curtains, wiping or cleaning them may affect the performance of the patented sporicidal formulation coated onto the curtain surface. The curtains are designed to kill pathogens in-situ and do not require any cleaning to improve their function. When any of our curtains become visibly soiled, they should always be replaced.
Can I leave Endurocide® Antimicrobial Plus Hospital Curtains in-situ when disinfecting with a misting/fogging machine?
No: it is recommended that Endurocide® Antimicrobial Plus Curtains are taken down when disinfecting with a misting/fogging machine.
The peroxide used in fogging/misting operations may react with the patented formulation coated onto the curtain's surface and over time reduce the effectiveness against pathogens. The use of peroxide is also likely to affect the labels affixed to the curtains, resulting in a visible discoloration of the information on the curtain label.
Can I hang Endurocide® Antimicrobial Plus Curtains in areas of direct sunlight?
It is advised that Endurocide® Antimicrobial Plus Curtains are not placed in areas where they are exposed to prolonged direct sunlight.
Although the curtains will still be effective against pathogens, the polypropylene material the curtains are made from is not UV resistant and may suffer accelerated ageing as a result: this will lead to a loss in tensile strength and elasticity of the curtains which will become visible as “pilling”.
Pilling is caused by the fibres in the polypropylene becoming brittle with UV exposure, breaking down and coming loose from the main fibre chains. This effect is normally only observed after several months in direct sunlight.
Any timescales provided/referenced are always offered as a guideline only and under no circumstances whatsoever constitute a guarantee.
Will Endurocide® Hospital Curtains work with existing tracking?
Endurocide® Antimicrobial Plus Curtains are available supplied with eyelets only or with a choice of seven different hanging systems so will fit work with most popular track systems.
- For further information on the Hanging Systems available, please refer to the Product Options tab.
Recycling & disposal of used Endurocide® Curtains
All parts of the Endurocide® Curtain, from the polypropylene fabric, through to the eyelets & hooks used on the curtain are made from 100% recyclable material and therefore Endurocide® Curtains are completely recyclable.
However, as the curtains are designed to come in contact with potential hospital pathogens and could be exposed to bodily fluids, actual disposal of curtains can take different forms depending on individual hospital protocols and the local regulations in place in each territory.
In general, there are the following disposal streams:
1. Second-life products
If hospital protocols allow, the curtains can be fully recycled. Polypropylene is a versatile material that can be re-used & re-purposed into second-life products. For example, we have some customers turning used curtains into plastic fences.
To the best of our understanding, recycling processes of polypropylene involve heating and melting which would destroy all potential pathogens on the curtains. However, please always check with your local recycling facility in your area for more information on what they could turn your curtains into.
2. Incineration
Should hospital protocols not allow the recycling of curtains that have come into contact with pathogens/bodily fluids, there is the possibility of incinerating the curtains after use for their calorific value. Polypropylene has a high calorific value and one full width curtain could potentially generate enough energy to light a standard light bulb for an hour and a half.
3. Hospital Waste
Should hospital protocols not allow the recycling of curtains and the hospital does not have the ability to incinerate curtains, Endurocide® Curtains can be disposed of as standard hospital waste in accordance with local protocols.
Should you have any questions about your individual hospital's needs, please contact us, or reach out to your local representative.
How often do Endurocide® Antimicrobial Plus Hospital Curtains need to be replaced?
Each hospital and sometimes individual wards (depending on the ward type and purpose) will have their own specific protocols and assessments regarding the changing of curtains and these should be adhered to.
However, Endurocide® Antimicrobial Plus Hospital Curtains have been independently tested and shown to offer antimicrobial & sporicidal properties for up to two years after manufacture. These test results have been demonstrated in both laboratory ‘box fresh’ test results, as well as independent peer review tests demonstrating real-life, in-situ testing by hospitals over a period of two years. For more information on these tests please click here, or contact us directly and our Technical Team will be happy to chat through any questions you may have.
Please note that Endurocide® Antimicrobial Plus Hospital Curtains are designed to be disposable and as such use polypropylene fabric. Should the polypropylene fabric become visibly dirty/soiled, we would recommend that the curtains should be changed, as they are not designed to be cleaned or washed.
Any timescales provided/referenced are always offered as a guideline only and under no circumstances constitutes a guarantee. The length of curtain hanging time will depend on individual ward use.
Have toxilogical studies been carried out on Endurocide® Antimicrobial Plus Hospital Curtains?
Yes: we have conducted an in-depth analysis of the individual chemical components present in our patented formulation which is then applied to our Endurocide® Antimicrobial Plus Curtains as a chemical blend during the treatment process.
To assess the risk of these individual chemical components, we have undertaken a technical analysis of the data available in the public domain and based assessments upon potential absorption and body mass calculations.
For the purposes of our assessments:
- We calculated that each standard, full width curtain is 15m2 (width: 7.5m x height: 2m). The Endurocide® treatment blend is uniformly applied to the curtain and equally distributed across the surface.
- We chose an area of the curtain approximately twice the surface area of the average hand (0.05m2 or 500cm2) and then calculated the mg/m2 of chemical blend used in this area.
- We then assessed the mg/m2 quantity of chemical blend in this single contact area for:
- Acute Toxicity risks
- Repeated Dose risks
- Calculations were based on average weighted UK males and females.
Conclusions:
| For acute exposure risk | We assessed both oral LD50 and dermal LD50 (chemical mg/kg body mass) and determined that there is not enough of any substance present in the single contact area to exceed the oral and dermal LD50 values. In all cases, the quantities of the components that we use in our formulation were significantly lower than the stated maximum LD50 limits. |
|
For repeated dose toxicity risk |
It was determined that none of the quantities of individual components used exceeded the “No observable Adverse Effect Level” limits. |
Glossary of Terms & Clarifications:
| Acute Toxicity | The adverse effects of a substance that results either from a single exposure or from multiple exposures in a short period of time (usually less than 24 hours.) |
| LD50 | Lethal dose (LD50) is the amount of an administered substance that kills 50 percent of a test group. It is typically expressed as milligrams of administered substance per kilogram of body mass. |
Are there any guarantees offered on Endurocide® Antimicrobial Plus Curtains?
No: whilst the biocidal technology used on Endurocide® Antimicrobial Plus Curtains has been independently tested and shown to have long lasting antimicrobial and sporicidal efficacy, the actual length of curtain use will depend on a variety of factors.
Such factors include, but are not limited to: individual hospital practices; the natural longevity of polypropylene; the risk of the curtain being soiled from items such as blood, urine and general spills; etc. Curtains should always be replaced when visibly soiled.
Any timescales provided/referenced are always offered as a guideline only and under no circumstances whatsoever constitute a guarantee.
Are Endurocide® Hospital Curtains fire retardant?
Yes: all types of Endurocide® Hospital Curtains.
Our Endurocide® Curtains have been independently tested and meet the following International Fire Retardant Standards:
- UK & Europe - BS 5867 Part 2 Types B&C: 2008
- USA & Canada - NFPA 701: 2010 and CAN/ULC-S109
- Australia & New Zealand - AS 2755.2-1985 and AS 1530.2-1993 Part 2
For copies of the certificates, please contact us.
Are Endurocide® Curtains available from the NHS Supply Chain?
Yes: a range of Endurocide® Antimicrobial Plus Hospital Curtains are available from the NHS Supply Chain in England and Wales.
- For further information and links to the NHS Supply Chain website, please click here.
What material are Endurocide® Hospital Curtains made from?
All our Endurocide® Curtains are manufactured from 100gsm polypropylene.
Polypropylene has a very low environmental impact when compared with other types of synthetic or traditional materials. In addition, the liquid used to impregnate our Endurocide® Antimicrobial Plus Curtains is water-based.
What pack sizes do Endurocide® Curtains come in?
Endurocide® Antimicrobial Plus Curtains are available in the following pack sizes:
|
|
Standard |
Long drop |
Meshtop |
Meshcut |
|
Case of 6 | Case of 5 | Case of 5 | Case of 5 |
|
Case of 8 | Case of 7 | Case of 7 | Case of 7 |
|
Case of 12 | Case of 10 | Case of 10 | Case of 10 |
- For further details on the product options of our curtains, please see the Product Options tab.
Are videos available of Endurocide® Curtains in use?
Yes: please see below for links to videos featuring our curtains used internationally -
Endurocide® Curtains help in the global fight against Coronavirus: Singapore
The below video shows the new COVID-19 Treatment Facility constructed at Singapore Expo using our Endurocide® Curtains throughout:
Endurocide® Curtains help in the global fight against Coronavirus: South Africa
The video below shows the South African president Cyril Ramaphosa touring one of the many ICU wards established recently across South Africa which were using our Endurocide® Curtains:
Please also visit our Youtube channel for additional videos and playlists.
Are brochures on the full range of Endurocide® products available to download?
How do I know I'm choosing the correct hook for my railing system?
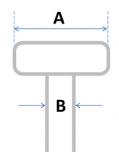
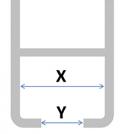
When choosing the type of hook you order with your curtain, it’s important that you order the correct size to fit your railing - and with so many types of railings available worldwide, this is sometimes easier said than done!
There are two ways to help ensure the hook will be suitable for your railing:
|
 |
||
|
 |
||
|
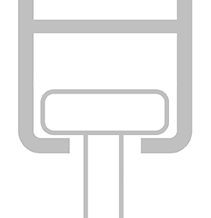 |
As a quick guide to our different types of hooks, please see the information below:
|
Hook Type |
Order Code |
Image |
Head width (A) |
Neck width (B) |
Suitable for railing system* |
|
QF |
|
10.0mm |
4.0mm |
Silent Gliss 6100 |
|
WH |
 |
9.5mm |
4.5mm |
Silent Gliss 6100 |
|
LT |
|
14.0mm |
3.0mm |
Movatrack 100 |
|
WWH |
|
12.0mm |
4.5mm |
Movatrack 100 |
|
MWW |  |
12.0mm | 4.5mm | Movatrack 100 |
|
U |
 |
N/A** |
N/A** |
Marlux Fast-Fit |
If you would like a sample pack of the different hooks we have available, please contact us.